The Basic Principles of Brick Carton Aseptic Packaging
The basic principle of aseptic packaging in paper boxes (such as the common brick-shaped carton and pillow-shaped pouch) can be summarized as follows: In a completely sterile environment, liquid food that has undergone ultra-high temperature (UHT) instantaneous sterilization is filled into pre-sterilized packaging materials and sealed to form a sterile container, thereby achieving long-term preservation of the food at room temperature.
The core lies in simultaneously achieving four conditions: "product sterility," "packaging material sterility," "filling environment sterility," and "sealing integrity." The essence of the entire process is "sterilization first, then filling."
The following is a detailed breakdown of its basic principles:
Core Process and Principle Diagram
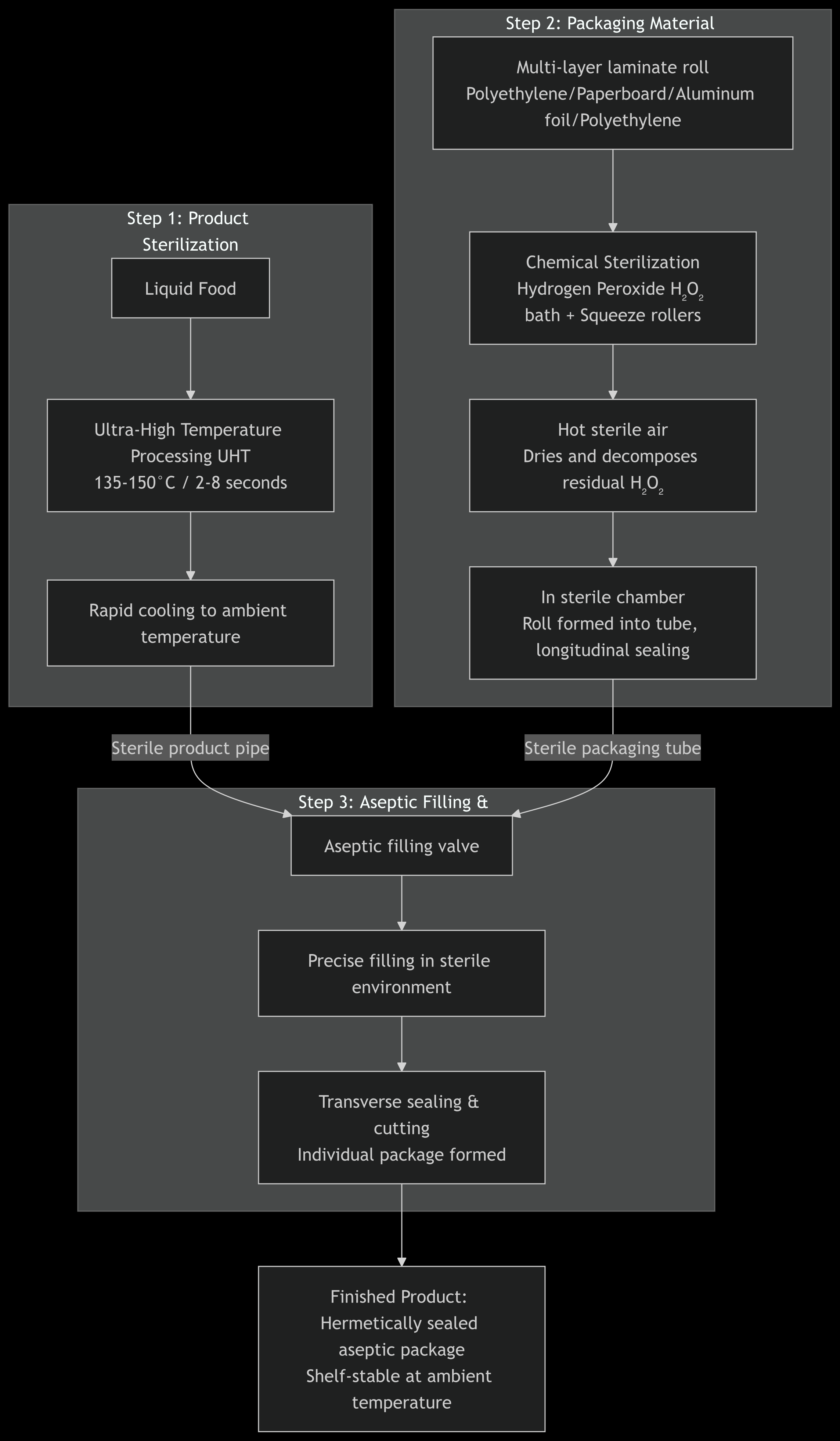
Detailed Explanation of Four Key Steps
1. Product Sterilization: Ultra-High Temperature (UHT) Instantaneous Sterilization
· Process: Liquid food (such as milk or juice) is heated to 135-150°C for a very short time (usually 2-8 seconds), and then rapidly cooled to room temperature (approximately 20-25°C).
• Purpose: To thoroughly kill all microorganisms (including bacteria and spores) and enzymes in the product, while preserving the original nutrients, flavor, and color of the food to the greatest extent possible due to the extremely short heating time.
• Result: A sterile product is obtained, ready for filling.
2. Sterilization of Packaging Materials: A combination of chemical and physical sterilization
• Materials: Cardboard packaging materials are in rolls and composed of multiple layers of composite materials (typically including polyethylene, cardboard, aluminum foil, etc.), effectively blocking light, oxygen, and microorganisms.
• Sterilization Process:
• Chemical Sterilization: The inner layer of the packaging material (the surface in contact with food) is thoroughly immersed in a hydrogen peroxide (H₂O₂) bath.
• Enhanced Sterilization: Immediately afterwards, it passes through a pair of heated extrusion rollers, which both squeeze out excess hydrogen peroxide and enhance the sterilization effect of the hydrogen peroxide with heat.
• Residue Removal: Finally, high-temperature sterile hot air or sterile air is sprayed onto the material surface to thoroughly dry and decompose any remaining hydrogen peroxide, breaking it down into harmless water vapor and oxygen. The inner surface of the packaging material reaches a commercially sterile state.
3. Creation and Maintenance of a Sterile Filling Environment
• Key Equipment: The entire filling process takes place in a closed, sterile chamber of a filling machine filled with sterile hot air or sterile air.
• Positive Pressure Protection: The air pressure inside the sterile chamber is always slightly higher than the external atmospheric pressure, ensuring that any possible non-sterile air cannot enter, forming an "air barrier."
• Filling Head: The filling head, which connects to the sterile product pipeline, also undergoes a rigorous sterilization process and remains under sterile protection throughout the production process.
4. Forming, Filling, and Sealing: Integrated Continuous Completion
• Forming: The sterilized packaging roll is folded into a tubular shape within the sterile chamber and then longitudinally sealed.
• Filling: Aseptic product is precisely injected through the filling head into the continuously descending packaging tube.
• Sealing: Below the liquid surface, the packaging tube is clamped by a pair of lateral sealing jaws, performing a lateral heat seal to seal the product within individual packages, while simultaneously completing the cutting. This seal is extremely strong, ensuring the integrity of the packaging.
Principle Summary and Advantages
The essence of aseptic packaging in cardboard boxes is "creating a locally sterile micro-world": sterilized food is placed into a freshly sterilized packaging container, completely isolated from the outside world. The entire process is completed instantaneously under continuous positive pressure sterile air protection.
The core advantages of this principle are:
• No cold chain required: Because the product, packaging, and environment are all sterile and perfectly sealed with strong barrier properties, the product can be stored at room temperature for months or even more than a year, significantly reducing logistics and storage costs.
• Maintaining quality: The combination of UHT technology and aseptic filling maximizes the preservation of the food's natural flavor and nutritional components.
• Lightweight Packaging: Packaging made of composite materials is lighter than glass jars and metal cans, making it easier to transport and carry.
• High Production Efficiency: The entire process is highly automated and continuous, resulting in high speed and efficiency.
In short, the brilliance of aseptic cardboard packaging lies in its approach: it doesn't rely on high-temperature sterilization after packaging (which would damage both the packaging and the food), but instead places the sterilization step beforehand, ensuring a sterile environment throughout the final sealing process, thus perfectly solving the preservation problem.